Unit Summary
This unit covers several key concepts in genetics. Mendels laws of inheritance including the law of segregation, law of dominance and the law of independent assortment are explained. It also explains the concepts of DNA, genes, chromosomes, alleles, genotype, phenotype, incomplete dominance, codominance, recessiveness, sex linkage and how they account for the transmission of hereditary characteristics according to Mendelian laws of inheritance. For further information on these, check the glossary at the bottom of the page.
The two phases of the cell cycle include interphase and mitosis. Phases of Mitosis include prophase, metaphase, anaphase and telophase. The similarities and differences between Mitosis and Meiosis are explained. The main difference is that meiosis contributes to genetic diversity in sexually reproducing organisms. This is due to the random assortment of tetrads, or crossing over during meiosis. The other differences include the following:
Genetic disorders can arise due to errors during replication of the genetic material, or during the separation of chromosomes during meiosis. Some examples of genetic disorders are Down Syndrome and Cystic Fibrosis. These genetic disorders can be detected by karyotyping, which is the number as well as the visual appearance in the cell nuclei of an organism.
The genetic material in our bodies is deoxyribonucleic acid, or DNA. DNA is packaged into chromatin, and chromatin condenses to form chromosomes. A duplicated chromosome consists of two sister chromatids. The DNA in chromatin is tightly coiled around proteins called histones. DNA consists of a long chain of subunits called nucleotides. Nucleotides have three parts:
Punnett squares are also used to predict the outcome of different genetic crosses, such as monohybrid and dihybrid cross. Students learned how to predict the likelyhood of the offspring having certain characteristics using Punnett squares. Students also learned about the ratios for each outcome, including the 9:3:3:1 phenotypic ratio. Blood types were also touched on during this unit.
Recent advancements in genetic technologies such as cloning, in vitro fertilization, genetic engineering, and artificial insemination are discussed. The use of prenatal screening techniques such as amniocentesis and chorionic villus sampling to detect genetic disorders in the unborn fetus are also discussed in this unit.
The two phases of the cell cycle include interphase and mitosis. Phases of Mitosis include prophase, metaphase, anaphase and telophase. The similarities and differences between Mitosis and Meiosis are explained. The main difference is that meiosis contributes to genetic diversity in sexually reproducing organisms. This is due to the random assortment of tetrads, or crossing over during meiosis. The other differences include the following:
- Meiosis is a form of sexual reproduction, mitosis is a form of asexual reproduction
- Meiosis occurs in humans, animals, plants, and fungi, while mitosis occurs in all organisms
- Meiosis produces 4 haploid cells, while mitosis produces 2 diploid cells
- In meiosis the chromosome number is reduced by half, while in mitosis the chromosome number remains the same
- Meiosis creates sex cells only, while mitosis creates all other cells
Genetic disorders can arise due to errors during replication of the genetic material, or during the separation of chromosomes during meiosis. Some examples of genetic disorders are Down Syndrome and Cystic Fibrosis. These genetic disorders can be detected by karyotyping, which is the number as well as the visual appearance in the cell nuclei of an organism.
The genetic material in our bodies is deoxyribonucleic acid, or DNA. DNA is packaged into chromatin, and chromatin condenses to form chromosomes. A duplicated chromosome consists of two sister chromatids. The DNA in chromatin is tightly coiled around proteins called histones. DNA consists of a long chain of subunits called nucleotides. Nucleotides have three parts:
- a phosphate group
- a ring shaped sugar called deoxyribose
- a nitrogenous base: a single or double ring of carbon and nitrogen atoms
Punnett squares are also used to predict the outcome of different genetic crosses, such as monohybrid and dihybrid cross. Students learned how to predict the likelyhood of the offspring having certain characteristics using Punnett squares. Students also learned about the ratios for each outcome, including the 9:3:3:1 phenotypic ratio. Blood types were also touched on during this unit.
Recent advancements in genetic technologies such as cloning, in vitro fertilization, genetic engineering, and artificial insemination are discussed. The use of prenatal screening techniques such as amniocentesis and chorionic villus sampling to detect genetic disorders in the unborn fetus are also discussed in this unit.
Baby Making Lab Report
| Genetic Processes Lab Report | |
| File Size: | 718 kb |
| File Type: | |
Genetic Disorder Project: Hemophilia
Hemophilia refers to a group of inherited bleeding disorders. There are three types of hemophilia: hemophilia A, hemophilia B, and hemophilia C. The most common type of hemophilia is type A, also known as classical hemophilia, accounting for about 80% of cases. Hemophilia B, also known as Christmas disease, accounts for about 20% of the cases, and hemophilia C, also known as Rosenthal Syndrome, is a very rare disorder. Hemophilia A is caused by a factor VIII deficiency, hemophilia B is caused by a factor IX deficiency, and hemophilia C is caused by factor XI deficiency. Factor VIII, IX, and XI are all coagulation factors that work as part of the blood clotting system. The frequency of hemophilia A is approximately 1 in every 5 000 to 10 000 people, whereas the frequency of hemophilia B is approximately 1 in every 50 000 people, and that of hemophilia C is approximately 1 in every 100 000 people.
The most common and recognizable symptoms of hemophilia are raised bruises on the stomach, back, chest, and butt, severe nosebleeds, hematuria (urinal bleeding), a child refusing to move muscles and joints, and severe bleeding subsequent to the following: surgery, loss of a tooth, or biting down on the lips or tongue. Hemophilia C is distinguished from A and B by the fact that it does not lead to bleeding in the joints. Hemophilia A and B are X linked recessive disorders and are more prevalent in males than females. An individual’s risk of developing hemophilia A or B is not affected by their race or ethnicity. Hemophilia C, unlike A and B, shows an autosomal recessive pattern of inheritance, and affects both males and females equally. Hemophilia C is more prevalent in Jews of Ashkenazi descent. Hemophilia is not a disorder that one develops throughout their life, but is expressed at birth. However, it is rarely noticed during the first six months of a baby’s life, as babies will not usually encounter incidents resulting in severe bleeding at this age. An individual is diagnosed with hemophilia after a series of tests are run. Simple blood tests can be performed to diagnose hemophilia. These tests include levels of coagulation factors: Factor VIII Level, Factor IX Level, and Factor XI Level. Additional tests include Prothrombin Time, Complete Blood Count, Partial Thromboplastin Time. Pregnant women can also undergo prenatal diagnosis, such as amniocentesis or chorionic villus sampling, to determine if the unborn child will develop hemophilia. Hemophilia A and B can be classified as mild, moderate, or severe depending on the amount of clotting factor in the blood. If the level of clotting factor VIII or IX in the blood is 5-30% of normal, it is classified as mild. If the levels are 1-5% of normal, it is classified as moderate, and if the levels are less than 1% of normal it is classified as severe hemophilia.
Hemophilia is a genetic disorder that impairs the human body’s ability to control bleeding as the blood does not clot normally. Blood clotting is essential to control and stop bleeding if a blood vessel is broken, therefore this disorder affects the circulatory system. Because of the frequent internal bleeding caused by this disorder, the blood vessels throughout the entire circulatory system are affected. Hemorrhaging creates mainly large bruises, however if the blood enters the joint spaces it can also cause swelling, leading to permanent damage to the joint and disfigurement. Hemophilia can also affect soft tissues in the body, such as muscular tissues. This can cause damage to the tissues and result in chronic debilitating condition, and would eventually require treatment. Depending on the location, hemorrhaging can have serious consequences and may be fatal, for example intracerebral hemorrhage.
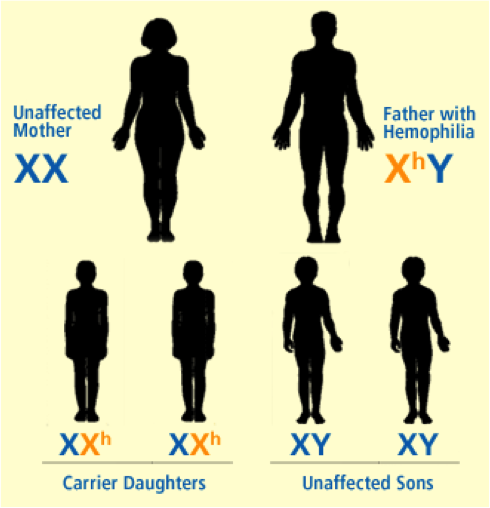
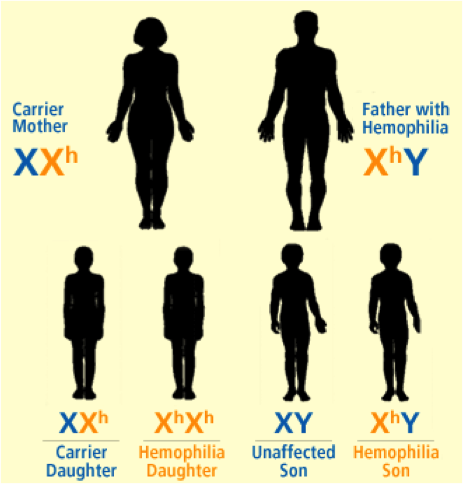
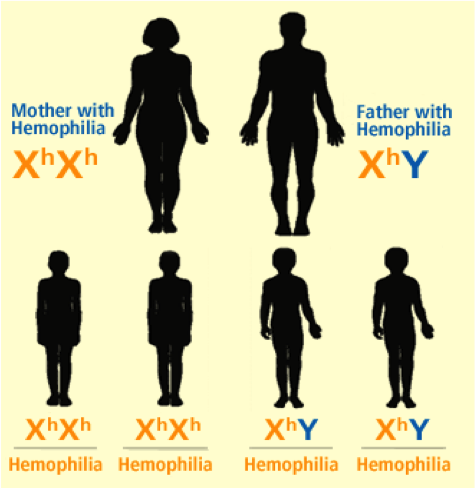
Hemophilia A and B are inherited in an X-linked recessive pattern, and the genes associated with this disorder are located on the X chromosome, therefore predominantly affecting males rather than females. This is explained by the fact that a male has one X chromosome and one Y chromosome, while females have two X chromosomes. If a female were to inherit one X chromosome with the mutant gene, she will be a carrier of the disorder, as she will still have a normal copy of the gene on the other X chromosome. The only way a female is affected by this disorder is if she inherits two X chromosomes, with a mutation in both copies of the gene. However, since males have only one X chromosome, if the X chromosome they inherit has the abnormal gene present on it, they will get the disorder. Figures 1-5 below show the possible inheritance patterns for the X-linked hemophilia A and B disorders.
The most common and recognizable symptoms of hemophilia are raised bruises on the stomach, back, chest, and butt, severe nosebleeds, hematuria (urinal bleeding), a child refusing to move muscles and joints, and severe bleeding subsequent to the following: surgery, loss of a tooth, or biting down on the lips or tongue. Hemophilia C is distinguished from A and B by the fact that it does not lead to bleeding in the joints. Hemophilia A and B are X linked recessive disorders and are more prevalent in males than females. An individual’s risk of developing hemophilia A or B is not affected by their race or ethnicity. Hemophilia C, unlike A and B, shows an autosomal recessive pattern of inheritance, and affects both males and females equally. Hemophilia C is more prevalent in Jews of Ashkenazi descent. Hemophilia is not a disorder that one develops throughout their life, but is expressed at birth. However, it is rarely noticed during the first six months of a baby’s life, as babies will not usually encounter incidents resulting in severe bleeding at this age. An individual is diagnosed with hemophilia after a series of tests are run. Simple blood tests can be performed to diagnose hemophilia. These tests include levels of coagulation factors: Factor VIII Level, Factor IX Level, and Factor XI Level. Additional tests include Prothrombin Time, Complete Blood Count, Partial Thromboplastin Time. Pregnant women can also undergo prenatal diagnosis, such as amniocentesis or chorionic villus sampling, to determine if the unborn child will develop hemophilia. Hemophilia A and B can be classified as mild, moderate, or severe depending on the amount of clotting factor in the blood. If the level of clotting factor VIII or IX in the blood is 5-30% of normal, it is classified as mild. If the levels are 1-5% of normal, it is classified as moderate, and if the levels are less than 1% of normal it is classified as severe hemophilia.
Hemophilia is a genetic disorder that impairs the human body’s ability to control bleeding as the blood does not clot normally. Blood clotting is essential to control and stop bleeding if a blood vessel is broken, therefore this disorder affects the circulatory system. Because of the frequent internal bleeding caused by this disorder, the blood vessels throughout the entire circulatory system are affected. Hemorrhaging creates mainly large bruises, however if the blood enters the joint spaces it can also cause swelling, leading to permanent damage to the joint and disfigurement. Hemophilia can also affect soft tissues in the body, such as muscular tissues. This can cause damage to the tissues and result in chronic debilitating condition, and would eventually require treatment. Depending on the location, hemorrhaging can have serious consequences and may be fatal, for example intracerebral hemorrhage.
Hemophilia A and B are inherited in an X-linked recessive pattern, and the genes associated with this disorder are located on the X chromosome, therefore predominantly affecting males rather than females. This is explained by the fact that a male has one X chromosome and one Y chromosome, while females have two X chromosomes. If a female were to inherit one X chromosome with the mutant gene, she will be a carrier of the disorder, as she will still have a normal copy of the gene on the other X chromosome. The only way a female is affected by this disorder is if she inherits two X chromosomes, with a mutation in both copies of the gene. However, since males have only one X chromosome, if the X chromosome they inherit has the abnormal gene present on it, they will get the disorder. Figures 1-5 below show the possible inheritance patterns for the X-linked hemophilia A and B disorders.
Changes, or mutations, in the F8 gene which codes for factor VIII are responsible for hemophilia A, whereas mutations in F9 gene which codes for the factor IX protein are responsible for hemophilia B. These mutations can result in an abnormal version of the protein or result in a reduced amount of one of these proteins, therefore these proteins cannot effectively participate in the blood clotting process. The gene locus for hemophilia A is Xq28, for hemophilia B is Xq27.1-27.2, and for hemophilia C is 4q35.
Currently, there is no cure for hemophilia. The only hope to cure hemophilia would be through gene therapy where the faulty gene is repaired or replaced. However patients who start prophylaxis early on in life (mean age of 3 years) show better musculoskeletal outcome with fewer joint bleeds. Without adequate treatment, individuals with hemophilia often die before even reaching adulthood. The average life expectancy for individuals who do receive proper treatments for hemophilia is approximately 10 years less than the life expectancy of a normal individual. Hemophilia has a large affect on the lives of individuals as even minor cuts or injuries must be closely monitored and treated if severe bleeding persists. Individuals with hemophilia often need to avoid any contact sports as these can lead to injuries involving bleeding. Most people with hemophilia can manage their condition and lead normal lives. The only cure for hemophilia is a liver transplant, however this often imposes much greater problems in an individual than hemophilia itself.
In the past, treatments for hemophilia included replacement therapy with patients receiving whole blood or plasma infusions to counteract severe bleeding. However, this was not very affective as the severe bleeding was only partly treated as it was not possible for factors VIII (lacking in hemophilia A) and IX (lacking in hemophilia B) to reach and sustain the levels necessary for effective blood coagulation. Cryoprecipitates made through cold precipitation of frozen plasma was the first effective treatment for hemophilia and this meant that treatment could be done at home and could be used as required. A large number of patients, however, became infected with blood bone pathogens such as Hepatitis C and HIV. This lead to the development of recombinant products, such as recombinant factor VIII and recombinant factor IX, which are free of all exogenous human and animal proteins, therefore posing minimal risk to the patient.
Currently, biotechnologists are trying to come up with a way that gene therapy can be used to treat hemophilia. The goal is to replace the diseased gene with a corrected version, with hopes of curing hemophilia. The ambitious objective of gene therapy is to edit a defective gene sequence in situ to achieve complete reversion of a disease phenotype for the lifetime of the patient. In spite of recent successes in site-specific correction of defective gene sequences, the focus of most gene therapy strategies to date is on gene addition rather than gene replacement. This simplified approach relies on a delivery mechanism to provide a corrected copy of the defective gene without removal of the error-containing genomic sequence. [1] One study was done in mice where genetically engineered Adeno Associated Virus (AAV) carrying the gene for coagulation factor was used to target the liver cells where these coagulation factors are normally produced. Mice that received the viral vector containing the gene were able to produce enough coagulation factor to reduce the blood clotting times to near normal levels. Control mice did not show significant improvements in clotting factor levels or clotting times, suggesting that this could be a promising approach. Clinical trials in humans using a similar approach are currently ongoing.[2]
Currently, there is no cure for hemophilia. The only hope to cure hemophilia would be through gene therapy where the faulty gene is repaired or replaced. However patients who start prophylaxis early on in life (mean age of 3 years) show better musculoskeletal outcome with fewer joint bleeds. Without adequate treatment, individuals with hemophilia often die before even reaching adulthood. The average life expectancy for individuals who do receive proper treatments for hemophilia is approximately 10 years less than the life expectancy of a normal individual. Hemophilia has a large affect on the lives of individuals as even minor cuts or injuries must be closely monitored and treated if severe bleeding persists. Individuals with hemophilia often need to avoid any contact sports as these can lead to injuries involving bleeding. Most people with hemophilia can manage their condition and lead normal lives. The only cure for hemophilia is a liver transplant, however this often imposes much greater problems in an individual than hemophilia itself.
In the past, treatments for hemophilia included replacement therapy with patients receiving whole blood or plasma infusions to counteract severe bleeding. However, this was not very affective as the severe bleeding was only partly treated as it was not possible for factors VIII (lacking in hemophilia A) and IX (lacking in hemophilia B) to reach and sustain the levels necessary for effective blood coagulation. Cryoprecipitates made through cold precipitation of frozen plasma was the first effective treatment for hemophilia and this meant that treatment could be done at home and could be used as required. A large number of patients, however, became infected with blood bone pathogens such as Hepatitis C and HIV. This lead to the development of recombinant products, such as recombinant factor VIII and recombinant factor IX, which are free of all exogenous human and animal proteins, therefore posing minimal risk to the patient.
Currently, biotechnologists are trying to come up with a way that gene therapy can be used to treat hemophilia. The goal is to replace the diseased gene with a corrected version, with hopes of curing hemophilia. The ambitious objective of gene therapy is to edit a defective gene sequence in situ to achieve complete reversion of a disease phenotype for the lifetime of the patient. In spite of recent successes in site-specific correction of defective gene sequences, the focus of most gene therapy strategies to date is on gene addition rather than gene replacement. This simplified approach relies on a delivery mechanism to provide a corrected copy of the defective gene without removal of the error-containing genomic sequence. [1] One study was done in mice where genetically engineered Adeno Associated Virus (AAV) carrying the gene for coagulation factor was used to target the liver cells where these coagulation factors are normally produced. Mice that received the viral vector containing the gene were able to produce enough coagulation factor to reduce the blood clotting times to near normal levels. Control mice did not show significant improvements in clotting factor levels or clotting times, suggesting that this could be a promising approach. Clinical trials in humans using a similar approach are currently ongoing.[2]
Footnotes:
[1] <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2408641/>.
[2] <http://clinicaltrials.gov/show/NCT01620801/>
Works Cited
Websites
1. Murphy, Samuel L., and Katherine A. High. "Abstract." National Center for Biotechnology Information. U.S. National Library of Medicine, 27 Aug. 2005. Web. 26 Mar. 2013. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2408641/>.
2. "TeensHealth." Hemophilia. KidsHealth, Web. 26 Mar. 2013. <http://kidshealth.org/teen/diseases_conditions/blood/hemophilia.html>.
3. Zaiden, Robert A. "Hemophilia A ." Hemophilia A. Web MD, Web. 26 Mar. 2013. <http://emedicine.medscape.com/article/779322-overview>.
4. "Bio Tech Spain." Hemophilia Treatment. Web 4 Bio, Web. 26 Mar. 2013. <http://biotechspain.com/en/news_item.cfm?iid=110627_noticia_genomeediting>.
5. "Hemophilia B Gene Therapy." Home. Clinical Trials, Oct. 2012. Web. 26 Mar. 2013. <http://clinicaltrials.gov/show/NCT01620801>.
Books
6. Freedman, Jeri. Hemophilia. New York: Rosen Pub. Group, 2007. Print.
7. Willett, Edward. Hemophilia. Berkeley Heights, NJ: Enslow, 2001. Print.
Articles
8. Press, Associated. "Biogen Seeks Approval of Hemophilia A Treatment." R & D Magazine, 13 Mar. 2013. Web. 26 Mar. 2013.
Glossary of Terms
Allele: The forms of a gene(different versions)
Amniocentesis - a medical procedure used in prenatal screening and diagnosis of chromosomal abnormalities
Carriers- heterozygous individuals who carry the recessive allele but are phenotypically normal (for example, Albinism).
Chromosome: one long, condensed DNA molecule containing hundreds or thousands of genes.
Codominance: an inheritance pattern in which neither allele is dominant to the other, both trait are expressed
Complete dominance- occurs when phenotypes of the heterozygote and dominant homozygote are identical.
Cross-fertilization: Two different True-breeding plants breeding
Crossing Over: Exchange of genetic material between homologous chromosomes
Cytokinesis: The division of the cytoplasm.
Dihybrid: created when crossing two true-breeding parents that differ in two character
DNA: Deoxyribonucleic Acid; A double-stranded that contains the genetic information for cell growth, division and function .
Dominant: allele that determines the trait that a heterozygous individual expresses.
Epistasis- a gene at one locus alters the phenotypic expression of a gene at a second locus.
F1-Generation (Fillial 1): P-Generation's offsprings
F2-Generation (Fillial 2): Product of when F1-Gernerations self-pollinate
Fertilization- the union of gametes (the sperm and the egg)
Gametophyte - The haploid stage of a plant life cycle that makes gametes (by mitosis)
Gene Locus: Site on a chromosome that a specific gene occupies
Gene: Unit of inherited information that carrys a code for a specific trait or function
Genetic Linkage: The tendency for alleles for different genes on the same chromosome to be inherited together
Genetic recombination - Production of new combination of alleles
Genetics- the scientific study of heredity and variation
Genome: full DNA sequence of an organism.
Genotype: an organism's genetic makeup
Heredity- the transmission of traits from one generation to the next
Heterozygous: Hybrid or mixed; Having different alleles at the same location in a chromosome, ex. Aa
Homozygous: True breed or pure; Having identical alleles on the same point in a chromosome, ex. AA
Incomplete dominance- the phenotype of F1 hybrids is somewhere between the phenotypes of the two parental varieties.
Karyotype: Display of all the choromosomes in a cell or individual
Law of independent assortment: Mendel's second law, which states that genes that segregate independently in meiosis do not influence each other's inheritance.
Law of segregation: Mendel's first law, which states that two alleles for a trait or gene separate during meiosis.
Lifecycle: Generation - generation sequence of stages in the reproductive history of an organism.
Meiosis - cell division to make sex cells (sperm and egg cells)
Mitotic Spindle: Is an apparatus of microtubules that controls chromosome movement during mitosis.
Monohybrids: Differ in one trait
Mutation: any change to the DNA of the cell.
Non-disjunction: Failure of homologous chromosomes to separate during anaphase I or II of meiosis, resulting in gametes with atypical numbers of chromosomes
Offspring: The product of reproduction, of a new organism produced by one or more parent(s).
Pedigree- a family tree that describes the interrelationships of parents and children across generations (gene history).
P-Generation(Parent): The true-breeding varieties
Phenotype: an organism's appearance
Pleiotrophy - when a single gene is affecting or multiple phenotypic traits
Polygenic: many genes that affect one trait
Punnet square: diagram that shows the expected proportions of all possible outcomes of a genetic cross.
Recessive: allele that is not expressed in a heterozygous individual.
Sporophyte - The diploid stage of a plant life cycle that makes haploid spores
Trait: Characteristics or attributes of an organism that are expressed by genes and/or influenced by the environment.
True-breeding: When a self-fertilized will produce identical offspring to itself generation after generation
Variation- the differences in appearance that offspring show from parents to siblings
Zygote- the fertilized egg (has one set of chromosomes from each parent)
Amniocentesis - a medical procedure used in prenatal screening and diagnosis of chromosomal abnormalities
Carriers- heterozygous individuals who carry the recessive allele but are phenotypically normal (for example, Albinism).
Chromosome: one long, condensed DNA molecule containing hundreds or thousands of genes.
Codominance: an inheritance pattern in which neither allele is dominant to the other, both trait are expressed
Complete dominance- occurs when phenotypes of the heterozygote and dominant homozygote are identical.
Cross-fertilization: Two different True-breeding plants breeding
Crossing Over: Exchange of genetic material between homologous chromosomes
Cytokinesis: The division of the cytoplasm.
Dihybrid: created when crossing two true-breeding parents that differ in two character
DNA: Deoxyribonucleic Acid; A double-stranded that contains the genetic information for cell growth, division and function .
Dominant: allele that determines the trait that a heterozygous individual expresses.
Epistasis- a gene at one locus alters the phenotypic expression of a gene at a second locus.
F1-Generation (Fillial 1): P-Generation's offsprings
F2-Generation (Fillial 2): Product of when F1-Gernerations self-pollinate
Fertilization- the union of gametes (the sperm and the egg)
Gametophyte - The haploid stage of a plant life cycle that makes gametes (by mitosis)
Gene Locus: Site on a chromosome that a specific gene occupies
Gene: Unit of inherited information that carrys a code for a specific trait or function
Genetic Linkage: The tendency for alleles for different genes on the same chromosome to be inherited together
Genetic recombination - Production of new combination of alleles
Genetics- the scientific study of heredity and variation
Genome: full DNA sequence of an organism.
Genotype: an organism's genetic makeup
Heredity- the transmission of traits from one generation to the next
Heterozygous: Hybrid or mixed; Having different alleles at the same location in a chromosome, ex. Aa
Homozygous: True breed or pure; Having identical alleles on the same point in a chromosome, ex. AA
Incomplete dominance- the phenotype of F1 hybrids is somewhere between the phenotypes of the two parental varieties.
Karyotype: Display of all the choromosomes in a cell or individual
Law of independent assortment: Mendel's second law, which states that genes that segregate independently in meiosis do not influence each other's inheritance.
Law of segregation: Mendel's first law, which states that two alleles for a trait or gene separate during meiosis.
Lifecycle: Generation - generation sequence of stages in the reproductive history of an organism.
Meiosis - cell division to make sex cells (sperm and egg cells)
Mitotic Spindle: Is an apparatus of microtubules that controls chromosome movement during mitosis.
Monohybrids: Differ in one trait
Mutation: any change to the DNA of the cell.
Non-disjunction: Failure of homologous chromosomes to separate during anaphase I or II of meiosis, resulting in gametes with atypical numbers of chromosomes
Offspring: The product of reproduction, of a new organism produced by one or more parent(s).
Pedigree- a family tree that describes the interrelationships of parents and children across generations (gene history).
P-Generation(Parent): The true-breeding varieties
Phenotype: an organism's appearance
Pleiotrophy - when a single gene is affecting or multiple phenotypic traits
Polygenic: many genes that affect one trait
Punnet square: diagram that shows the expected proportions of all possible outcomes of a genetic cross.
Recessive: allele that is not expressed in a heterozygous individual.
Sporophyte - The diploid stage of a plant life cycle that makes haploid spores
Trait: Characteristics or attributes of an organism that are expressed by genes and/or influenced by the environment.
True-breeding: When a self-fertilized will produce identical offspring to itself generation after generation
Variation- the differences in appearance that offspring show from parents to siblings
Zygote- the fertilized egg (has one set of chromosomes from each parent)